Fumed Silica Manufacturing Process & Morphology: A Technical Guide
Executive Summary
- What it is: Fumed silica is a synthetic, amorphous silicon dioxide (SiO2) with ultra-low bulk density and high specific surface area.
- How it's made: Produced via continuous flame hydrolysis of silicon tetrachloride (SiCl4) at temperatures exceeding 1500°C.
- Particle Growth: Evolves kinetically from primary particles (7-40 nm) to fused aggregates (100-500 nm), and finally to physical agglomerates (1-200 μm).
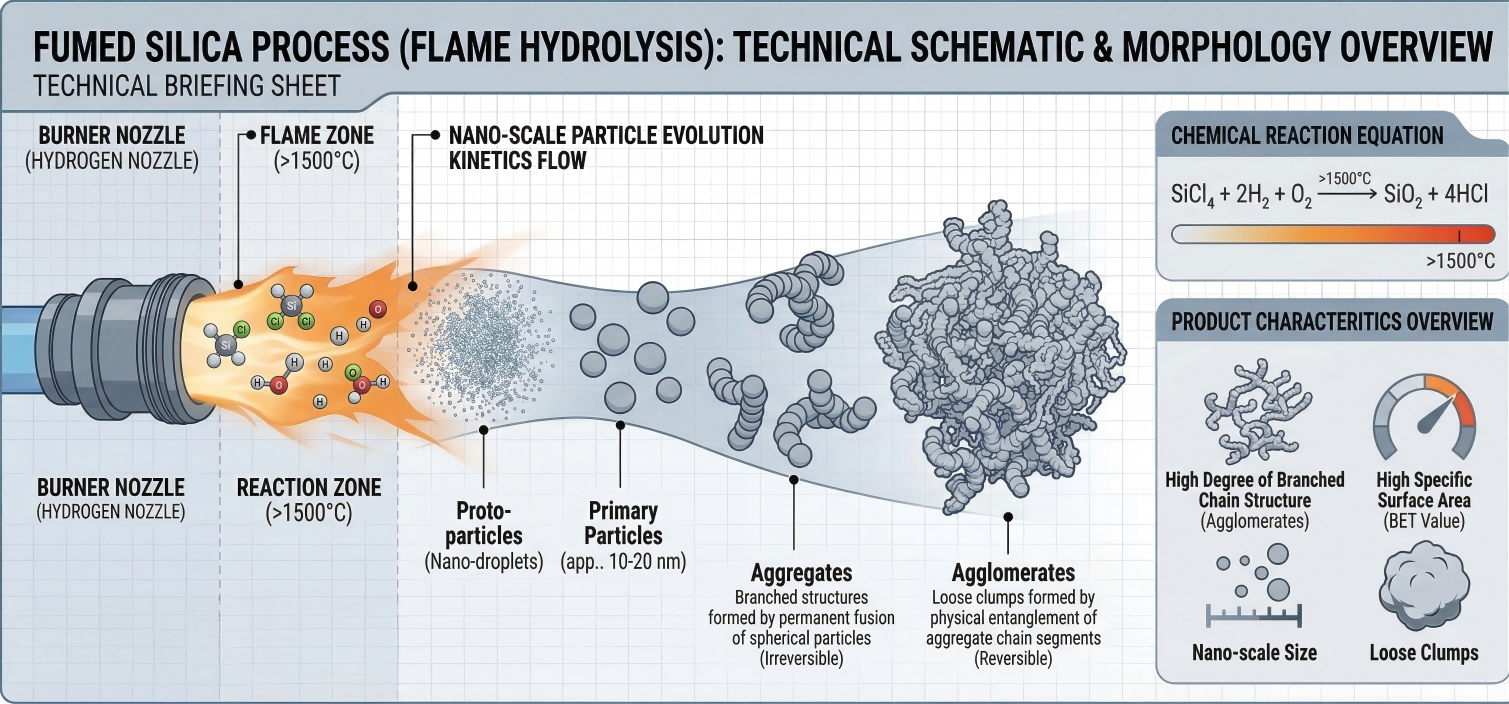
1. Flame Hydrolysis & Particle Evolution Kinetics
The core of fumed silica production takes place in a specialized burner reactor. Silicon tetrachloride (SiCl4) is vaporized and hydrolyzed in a hydrogen-oxygen flame. The stoichiometric reaction is defined as:
The ultimate performance of the silica depends entirely on the strict thermodynamic and kinetic control of particle growth, which occurs in three distinct stages:
Primary Particles
Precursor gases collide to form non-porous, spherical SiO2 nano-droplets within the flame core.
Fused Aggregates
Partially molten primary particles collide and irreversibly fuse into hard, three-dimensional branched chains.
Agglomerates
During cooling, aggregates physically entangle via hydrogen bonding and van der Waals forces into reversible floccs.
2. The Industrial Manufacturing Process
The continuous production of fumed silica is a highly controlled sequence involving high-temperature synthesis, gas-solid separation, and surface purging.
-
Step 1: Vaporization & Mixing
Liquid silicon tetrachloride is vaporized and precisely mixed with dry hydrogen and air to form a homogenous reactant gas.
-
Step 2: Flame Synthesis
The mixture is ignited in a combustion chamber. The resulting aerosol consists of solid SiO2 particles suspended in hydrochloric acid (HCl) gas.
-
Step 3: Gas-Solid Separation
The mixture passes through cooling sections and enters cyclones or bag filters, where the silica powder is separated from the acidic off-gas.
-
Step 4: Deacidification (Purging)
The separated silica passes through a calciner. Heated air or steam (400-600°C) is used to strip away residual HCl adsorbed on the silica surface, neutralizing the pH.
-
Step 5: Densification & Packaging
Freshly produced fumed silica has a bulk density of only 15-20 g/L. It undergoes vacuum densification (typically to 50 g/L) to facilitate logistics before packaging.
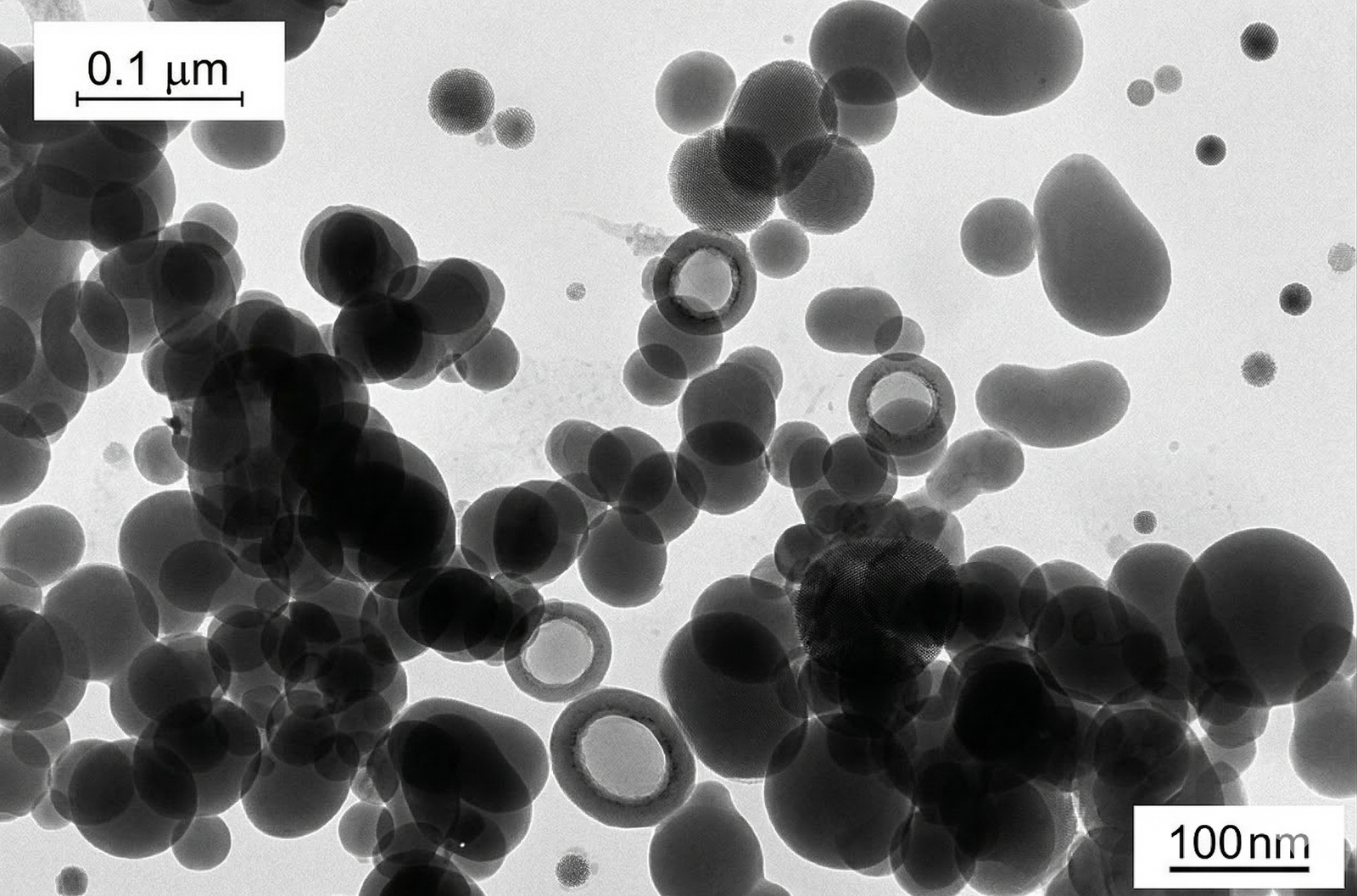
3. Morphological Characteristics: Macro to Micro
Macroscopically, fumed silica is an extremely fluffy, highly flowable white powder. However, its true functional power—providing structural support, thickening, and thixotropy—originates from its microscopic branched aggregate structure visible only under Transmission Electron Microscopy (TEM).